LECTURE
10
Polytropic
Process
W = ò cdv/ vn
w =
(P1v1- P2v2)/(n-1)
du =
dq – dw
u2
– u1 = q - (P1v1- P2v2)/(n-1)
u2
– u1 = Cv (T2
– T1) = q – w
q =
R(T2 – T1)/(g-1) + (P1v1- P2v2)/(n-1)
= R
(T1 – T2){1/(n-1) – 1/(g-1)}
=(P1v1-
P2v2)/(n-1) {(g -n)/(g-1)}
=w.{
(g -n)/(g-1)}
Problem: Air (ideal gas with g = 1.4) at 1 bar and 300K is compressed till
the final volume is one-sixteenth of the original volume, following a
polytropic process Pv1.25 = const. Calculate (a) the final pressure
and temperature of the air, (b) the
work done and (c) the energy transferred as heat per mole of the air.
Solution: (a) P1v11.25 = P2v21.25
P2
= P1(v1/v2)1.25 = 1(16)1.25
= 32 bar
T2
= (T1P2v2)/(P1v1) = (300
x 32 x 1)/(1x16)
= 600K
(b) w =
(P1v1- P2v2)/(n-1)
= Ru(T1 –
T2)/(n-1)
=
8.314 (300 – 600)/(1.25-1) = -9.977 kJ/mol
(c) q = w.{ (g
-n)/(g-1)}
= -9.977 (1.4 – 1.25)/(1.4-1)
= -3.742 kJ/mol
Unresisted or Free
expansion
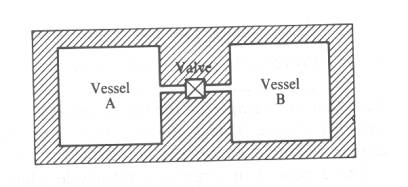
In
an irreversible process, w ¹ ò Pdv
Vessel
A: Filled with fluid at pressure
Vessel
B: Evacuated/low pressure fluid
Valve is
opened: Fluid in A expands and fills
both
vessels A and B. This is known as unresisted expansion or free expansion.
No
work is done on or by the fluid.
No
heat flows (Joule’s experiment) from the boundaries as they are insulated.
U2
= U1 (U = UA + UB)
Problem: A rigid and insulated container of 2m3
capacity is divided into two equal compartments by a membrane. One compartment
contains helium at 200kPa and 127oC while the second compartment
contains nitrogen at 400kPa and 227oC. The membrane is punctured and
the gases are allowed to mix. Determine the temperature and pressure after
equilibrium has been established. Consider helium and nitrogen as perfect gases
with their Cv as 3R/2 and
5R/2 respectively.
Solution:
Considering
the gases contained in both the compartments as the system, W= 0 and Q = 0.
Therefore, DU = 0 (U2 = U1)
Amount of helium = NHe
= PAVA/RuTA
= 200 x 103
x 1/(8.314 x400)
= 60.14 mol.
Amount of nitrogen =
NN2 = PBVB/RuTB
= 400 x 103
x 1/(8.314x500)
= 96.22 mol.
Let Tf be
the final temperature after equilibrium has been established. Then,
[NCv(Tf-400)]He
+ [NCv(Tf-500)]N2 = 0
Ru[60.14(Tf-400)3
+ 96.22(Tf-500)5 ] /2 = 0
Or, Tf = 472.73 K
The
final pressure of the mixture can be obtained by applying the equation of
state:
PfVf
= (NHe + NN2)Ru Tf
2Pf = (60.14 + 96.22) 8.314 (472.73)
or, Pf = 307.27 kPa