Fuels and Combustion
Most of the fuels contain Carbon and Hydrogen and are in solid, liquid or in gaseous form.
1.
Solid Fuels (Coal): Consists of moisture, volatile matter, fixed carbon
and ash. The analysis specifies on a mass basis, the relative amounts of these
constituents. The ultimate analysis may be given on the dry basis.
2.
Liquid Fuels: Most liquid fuels are mixtures of many different
Hydrocarbons. Common examples are Gasoline, Kerosene, Diesel oil etc. Commonly
a liquid fuel is treated as a single hydrocarbon with an empirical formula CxHy even though it is a mixture of several
hydro carbons.
3.
Gaseous fuels: Natural gas (mainly Methane), coal gas (a mixture of
methane and Hydrogen) etc.
In most of the
combustion processes the required Oxygen is not supplied as pure Oxygen but is
supplied as air. Air contains 21 mol
percent O2 and 79 mol percent of N2.
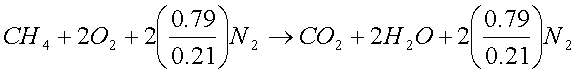
![]()
The minimum
amount of air which supplies the required amount of oxygen for complete
combustion of a fuel is called the stoichiometric or theoretical air.
The amount of air
in excess of the stoichiometric air is called excess air. It is usually
expressed in terms of the stoichiometric air as percent excess air.
Amount of air less than stoichiometric are called deficiency of air.
General combustion reaction
![]()
the coefficients
to the substances called stoichiometric coefficients. The balance of atoms
yields the theoretical amount of air as
C: ![]() = x
= x
H: 2![]() =y
=y
N2: ![]() =3.76*
=3.76* ![]()
O2: ![]()
And the total
number of moles of air for 1 mole of fuel becomes
nair =
![]()
Two important
parameters often used to express the ratio of fuel and air are the air-fuel
ratio ( AF ) and its reciprocal, the fuel-air ratio ( FA ) .
AFmass=
mair / mfuel
AFmole=
nair / nfuel
They are related through molecular weight as
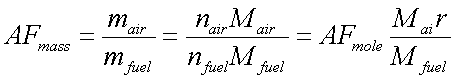
problem:
Methane (CH4)
is burned with atmospheric air. The analysis of the products on a dry basis is
as follows:
CO2 10.00%
O2 2.37
CO 0.53
N2 87.10
-----------
100.00%
Calculate the air-fuel ratio
and the percent theoretical air, and determine the combustion equation.
Solution:
The solution consists of
writing the combustion equation for 100 kmol of dry products, introducing
letter coefficients for the unknown quantities, and then solving for them.
From the analysis of the
products, the following equation can be written, keeping in mind that this
analysis is on a dry basis.
![]()
A balance for each of the elements will enable us to solve for all the unknown coeffiecients:
Nitrogen balance: c = 87.1
Since all the nitrogen comes
from the air,
c / b =3.76 b=87.1/3.76=23.16
Carbon balance: a = 10.00+0.53=10.53
Hydrogen balance: d=2a=21.06
Oxygen balance: All the
unknown coefficients have been solved for, and therefore the oxygen balance
provides a check on the accuracy. Thus, b can also be determined by an oxygen
balance.
b=10.00+0.53/2+2.37+21.06/2=23.16
substituting these values
for a,b,c and d we have
![]()
Dividing through by 10.53
yields the combustion equation per kmol of fuel.
![]()
The air-fuel ratio on a mole
basis is
2.2+8.27=10.47 kmol air/kmol
fuel
The fuel-air on a mass basis
is found by introducing the molecular weights.
AF=10.47*28.97/16.0=18.97 kg
air/kg fuel
The theoretical air-fuel
ratio is found by writing the combustion equation for theoretical air.
![]()
AFtheo=
(2+7.52)28.97/16.0=17.23 kg air/kg fuel
The percent theoretical air
is 18.97/17.23= 110%
Problem
A particular coal has the
following ultimate analysis on a dry basis, percent by mass:
Component |
Percent by mass |
|
Sulfur |
0.6 |
Hydrogen |
5.7 |
|
Carbon |
79.2 |
|
Oxygen |
10.0 |
|
Nitrogen |
1.5 |
|
Ash |
3.0 |
This coal is to be burned
with 30% excess air. Calculate the air-fuel ratio on a mass basis.
Solution:
One approach to this problem
is to write the combustion equation for each of the combustible elements per
100 kg of fuel. The molar composition per 100 kg of fuel is found first.
Kmol S / 100 kg fuel =
0.6/32 = 0.02
Kmol H2 / 100 kg
fuel = 5.7/2 = 2.85
Kmol C / 100 kg
fuel=79.2/12=6.60
Kmol O2 / 100 kg
fuel=10/32=0.31
Kmol N2 / 100 kg
fuel=1.5/28=0.05
The combustion equations for
the combustible elements are now written, which enables us to find the
theoretical oxygen required.
![]()
![]()
![]()
8.04 kmol O2
required / 100 kg fuel
-0.31 kmol O2 in
fuel / 100 kg fuel
7.73 kmol O2 from
air / 100 kg fuel
AFtheo=[7.73+7.73(3.76)]28.97/100=10.63
kg air/kg fuel
For 30%excess air the
air-fuel ratio is
AF=1.3*10.63=13.82 kg air/
kg fuel