Irreversibility
The irreversibility of a process is defined as the difference between the useful work and the actual work
Consider the system ant it surroundings as a compositive system, the work done by the composite system under adiabatic condition
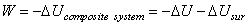 |
|
or 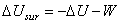 |
(26.17) |
Application of the two laws of thermodynamics to the surroundings gives
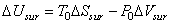
|
(26.18) |
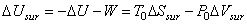 |
(26.19) |
Substituting (26.19) in (26.16)
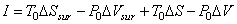
|
(26.18) |
or 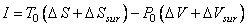 |
(26.20) |
Net change in the volume of the composite system is zero
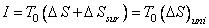 |
(26.21) |
For an irreversible process 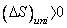 and for a reversible process, and for a reversible process, 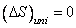 . .
Irreversibility (Alternative Method)
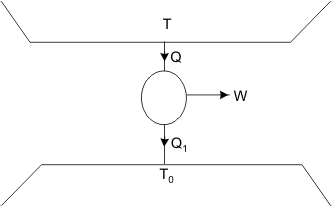
Figure 26.2
Refer to Figure 26.2
|