Compressibility Chart
To quantify deviation of real behavior from the ideal gas behavior, we introduce a new term namely, the compressibility factor. The compressibility factor Z is defined as the ratio of the actual volume to the volume predicted by the ideal gas law at a given temperature and pressure.
Z = (Actual volume) / (volume predicted by the ideal gas law)
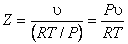 |
(10.10) |
If the gas behaves like an ideal gas, Z =1 at all temperatures and pressures. A plot of Z as a function of temperature and pressure should reveal the extent of deviation from the ideal gas law. Figure 10.1 shows a plot of Z as a function of temperature and pressure for N2.

Figure 10.1
For each substance, a compressibility factor chart or compressibility chart is available.

Figure 10.2 It would be very convenient if one chart could be used for all substances. The general shapes of the vapour dome and of the constant temperature lines on the  plane are similar. plane are similar.
This similarity can be exploited by using dimensionless properties called reduced properties. The reduced pressure is the ratio of the existing pressure to the critical pressure of the substance and the same is followed for the reduced temperature and reduced volume. Then
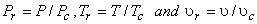 |
(10.11) |
At the same temperature and pressure, the molar volumes of different gases are different. However, it is found from experimental data that at the same reduced pressure and reduced temperature, the reduced volumes of different gases are approximately the same.
Therefore for all substances
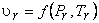 |
(10.12) |
or,
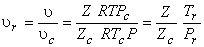 |
(10.13) |
Where 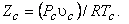  is called critical compressibility factor. Experimental values of is called critical compressibility factor. Experimental values of  for most substance fall with in a narrow range 0.2-0.3. Therefore from the above equation we can write for most substance fall with in a narrow range 0.2-0.3. Therefore from the above equation we can write
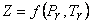 |
(10.14) |
When Z is plotted as a function of reduced pressure and 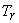 , a single plot, known as general compressibility chart (Figure 10.2) is found to be satisfactory for a wide variety of substances. Although necessarily approximate, the plots are extremely useful in situations where detailed data on a particular gas are lacking but its critical properties are available. , a single plot, known as general compressibility chart (Figure 10.2) is found to be satisfactory for a wide variety of substances. Although necessarily approximate, the plots are extremely useful in situations where detailed data on a particular gas are lacking but its critical properties are available.
|