Research
Materials Chemistry
Materials Chemistry
The design and development of new material are indispensable for sustainable growth and is at the heart of chemistry. Though we are not specialists in this area, we try to contribute to this by understanding the material's photophysics and subsequently predicting suitable modifications to have the desired function. We are specifically interested in providing this physical insight.
Current research:
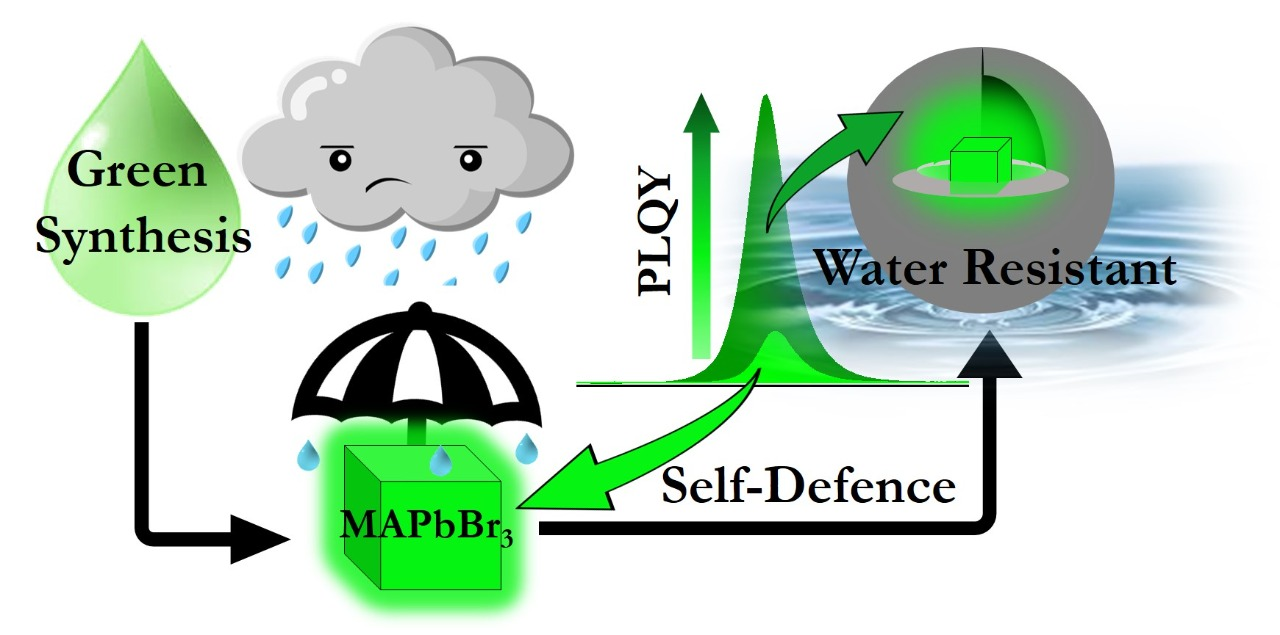
- Inspired by our understanding of the solution chemistry, we are now trying to synthesize photoluminescence, water-stable perovskite nanocrystal highly in a green medium.
- Synthetic and post-synthetic modifications to have better nanocrystals.
- Contemplating the charge carrier dynamics to understand the physical basis of doping.
Contact:
Spectroscopic insights into perovskite nanocrystals
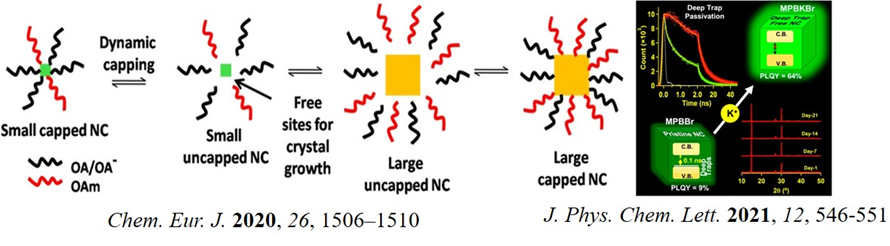
The last decade saw fast growth in the field of perovskite nanocrystal for high-efficiency optoelectronic applications. However, several issues prevent the use of this material for practical technology. Some of these issues are low moisture stability, associated potential environmental hazards, low thermal stability, and lower quantum yield (when some other ions substitute pb2+). In our laboratory, we are mainly working on the development and synthesis of organic-inorganic hybrid lead halide perovskite nanocrystal systems. More importantly, as a physical chemist, we intend to understand the photophysics and charge carrier dynamics of such materials. Recent studies on Bi3+-doped perovskites materials contributed as a new strategy of developing perovskite materials. Despite better stability and greener character, Bi3+-doped perovskites are inferior to pristine lead analogue due to weak photoluminescence. We investigated the molecular origin of this. We proposed that it is due to the ultrafast deep trapping transfer and not due to surface traps. Expectedly, we increased the photoluminescence quantum yield enormously by the passivation of these deep trap states by K+ (J. Phys. Chem. Lett. 2021, 12, 546). We also explained the reversible ultra-slow crystal growth of Bi3+-doped Pb perovskite by dynamic capping (Chem. Eur. J. 2020, 26, 1506).
Selective and sensitive chemo-sensing of various biologically important ions
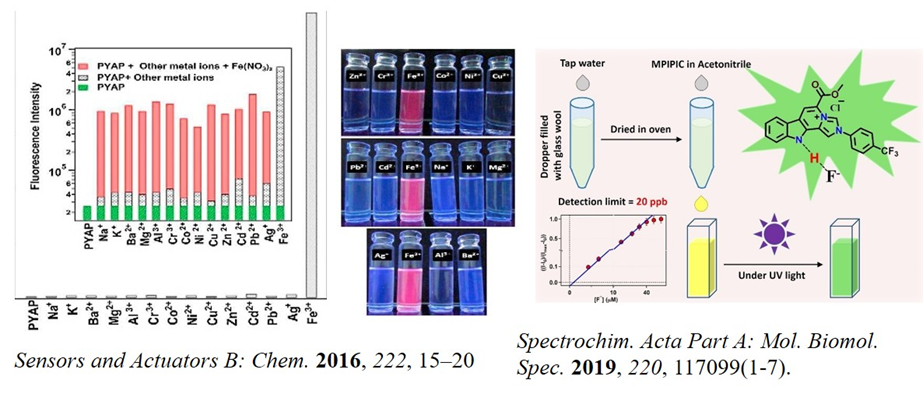
Various cations and anions are essential in the physiological process of the living cell. An optimum concentration of these ions is necessary to maintain health. The optimum concentrations for various ions are in the ppm level. Therefore, a fast, selective, and sensitive detection of these ions in aqueous solution at the sub-ppm level is very important. We synthesized some organic ligands that can selectively and sensitively sense various ions, including Cu2+ (Curr. Anal. Chem. 2021; ChemistrySelect 2020, 5, 9435; Inorg. Chem. 2012, 51, 8664), Fe3+(Sensors and Actuators B: Chem. 2016, 222, 15), and F- (Spectrochim. Acta Part A: Mol. Biomol. Spec. 2019, 220, 117099).

