|
Bioinorganic Laboratory | ||||||
|
|
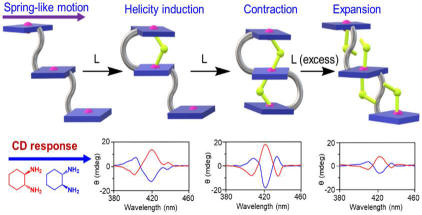
Selected Publications17. Modulation of Supramolecular Chirality by Stepwise Axial Coordination in a Nano-size Trizinc(II)porphyrin Trimer A. Dhamija, D. Chandel and S. P. Rath* Chem. Sci., 2023, 14, 6032-6038.Full Article (Edge Article)
The work
demonstrates unique chiral guest’s triggered spring-like contraction and
extension motions coupled with unidirectional twisting upon successive
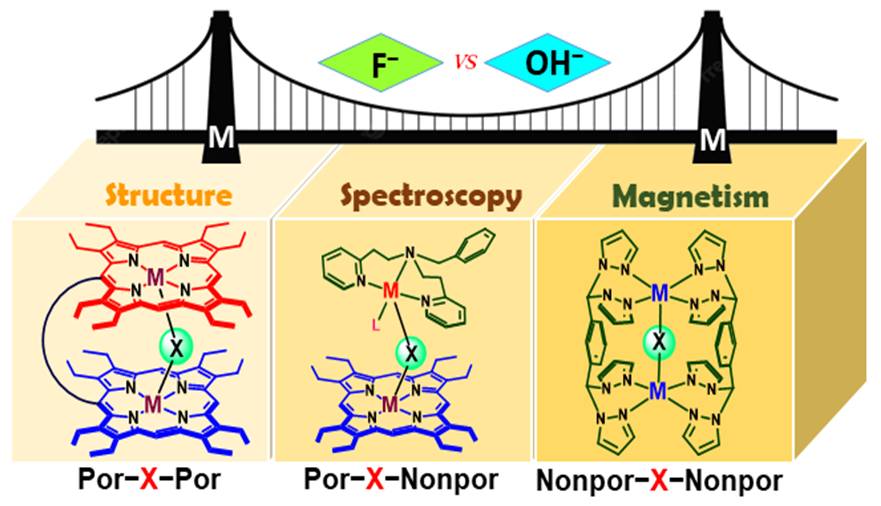
coordination to a flexible and ‘nano-size’ achiral 16. Binuclear Complexes with Single M-F-M Bridge (M: Fe, Mn, and Cu): A Critical Analysis of the Impact of Fluoride for Isoelectronic Hydroxide Substitution S. Sarkar, F. S. T. Khan, T. Guchhait and S. P. Rath* Coord. Chem. Rev. 2023, 479, 215003. Full Article
In this review article, we describe an
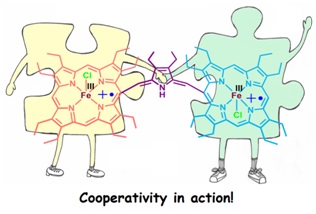
up-to-date progress of single fluoro-bridged dinuclear metal complexes 15. Cooperativity in Diiron(III)porphyrin Dication Diradical-Catalyzed Oxa-Diels-Alder Reactions: Spectroscopic and Mechanistic Insights S. Sarkar, P. Sarkar, D. Samanta, S. K. Pati and S. P. Rath* ACS Catal. 2022, 12, 9589-9601. Full Article
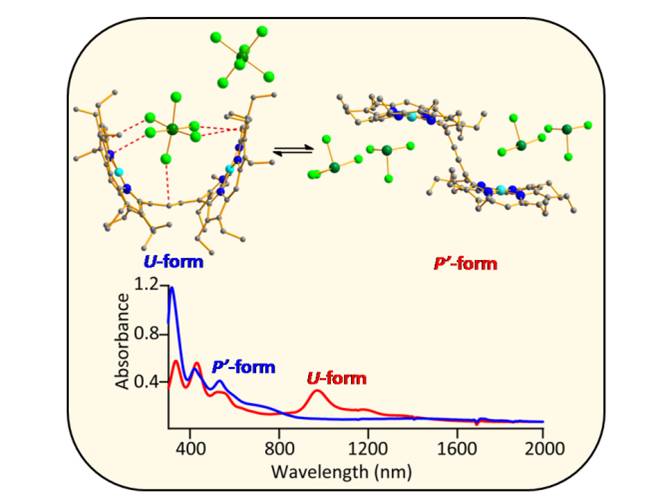
A dication diradical diiron(III) porphyrin dimer was devised as a competent catalyst for the oxa-Diels Alder (ODA) type reaction of aldehydes with 1,3-dienes. The computational studies corroborated the detailed spectroscopic investigation which bespoke of the pivotal roles played by the metal ion as the Lewis acidic centers and also enabled us to delve deeper into the reaction mechanism. The efficacy of employing such a diheme catalyst over mono heme analog is manifested in the cooperative effect which resulted in lower catalyst loading with excellent yields. 14. Counter Ion Triggers Stabilization of Two Geometrical Isomers of a Ni(II)dication Diradical Porphyrin Dimer: Role of Anion-π Interactions A. K. Pandey. M. Usman and S. P. Rath* Chem. Commun. 2019, 55, 7926-7929. Full Article
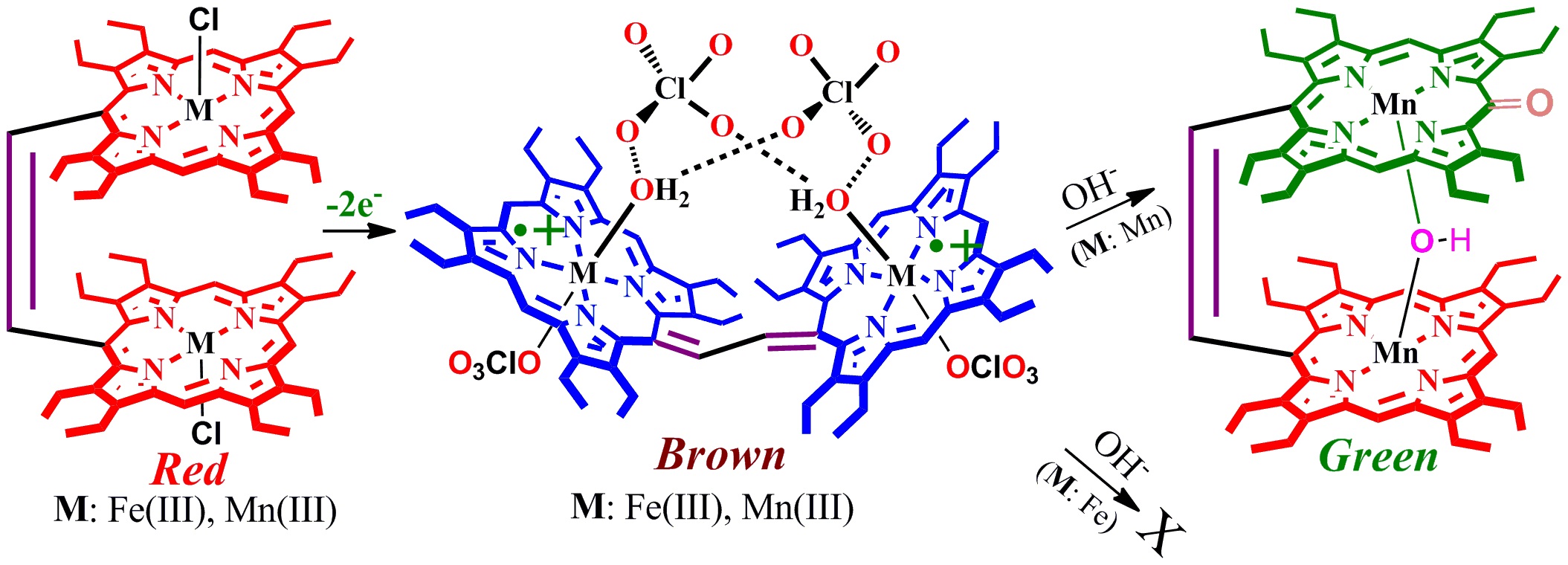
Two geometrical isomers (namely U and P'-forms) of nickel(II) dication diradical porphyrin dimer have been isolated selectively in pure form just by varying oxidant concentrations in the same reaction mixture. Both isomers, stabilized exclusively by anion-π interactions, have unique and very distinct electronic and spectroscopic features and display anion-induced charge/electron transfer phenomenon. 13. A Dimanganese(III) porphyrin dication diradical and its transformation to a μ-hydroxo porphyrin-oxophlorin heterodimer A. Kumar, D. Sil, M. Usman and S. P. Rath* Chem. Commun. 2019, 55, 1588-1591. Full Article
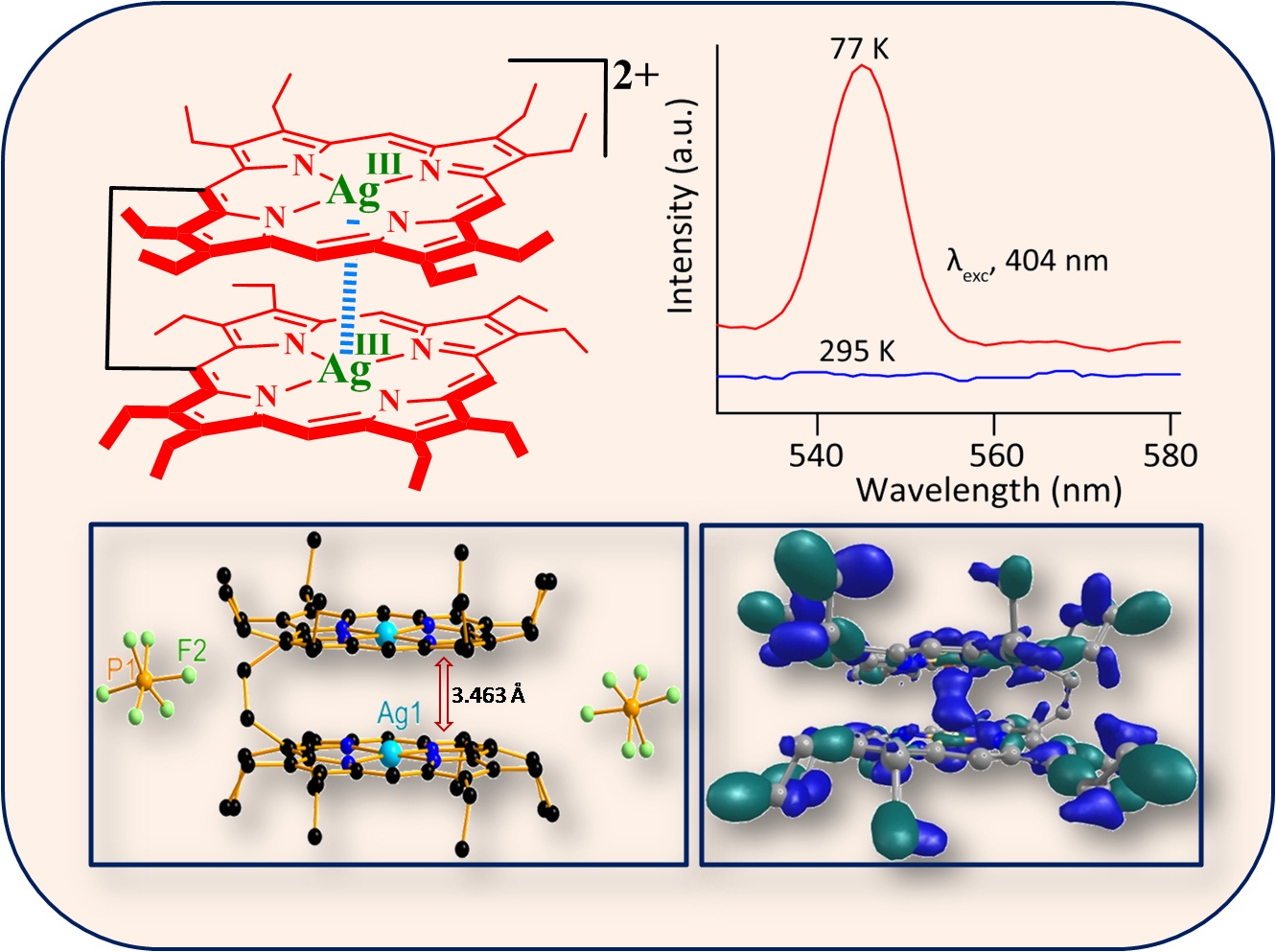
Spin coupling models are different between iron and manganese in their dication diradical complexes which reflected in their reactivity: while addition of tetrabutylammonium hydroxide to dimanganese(III) dication diradical complex quickly converts into the μ-hydroxo dimanganese(III) porphyrin-oxophlorin heterodimer, its dii-iron(III) analog remains very stable. 12. Silver(III)···Silver(III) Interaction that Stabilize the syn Form in a Porphyrin Dimer Upon Oxidation A. K. Singh, F. S. T. Khan and S. P. Rath* Angew. Chem. Int. Ed. 2017, 56, 8849-8854. Full Article
Meeting at the bridge: Ethane-bridged silver(III) porphyrin dimer exhibits significant metallophilic interaction that brings two porphyrin rings on top of each other with short Ag(III)···Ag(III) distance attributed to the overlap of dz2 metal orbital. The complex displays an intense emission peak at 546 nm at 77 K in the photoluminescence study. 11. Oxo- and hydroxo-bridged diiron(III) porphyrin dimers: inorganic and bio-inorganic perspectives and effects of intermacrocyclic interactions T. Guchhait, S. Sasmal, F. S. T. Khan and S. P. Rath * Coord. Chem. Rev. 2017, 337, 112-144. Full Article
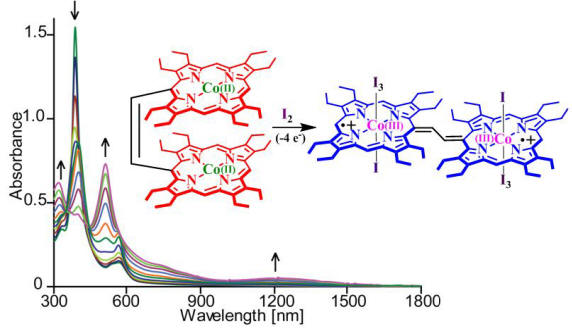
The fascinating chemistry of the oxo and hydroxo-bridged diheme centers, covalently connected through bridges varying in lengths and flexibility, has attracted much attention in recent times and a comprehensive review on their syntheses, structures and spectral properties is presented here. 10. A Highly Oxidized Cobalt Porphyrin Dimer: Spin Coupling and Stabilization of the Four-Electron Oxidation Product S. Dey, D. Sil and S. P. Rath* Angew. Chem. Int. Ed. 2016, 55, 996-1000. Full Article
Four to be sure: The oxidation of a cobalt(II) porphyrin dimer with iodine readily produced a cobalt(III) porphyrin π-cation radical dimer. Extensive π conjugation through the ethylene bridge led to several unusual spectral and geometrical features of this four-electron oxidized complex and stabilized the singlet state by enabling strong antiferromagnetic coupling between the π-cation radicals. 9. Oxidation Triggers Extensive Conjugation and Unusual Stabilization of Two Di-heme Dication Diradical Intermediates: Role of Bridging Group for Electronic Communication D. Sil, S. Dey, A. Kumar, S. Bhowmik and S. P. Rath* Chem. Sci. 2016, 7, 1212-1223. (Edge Article) Full Article
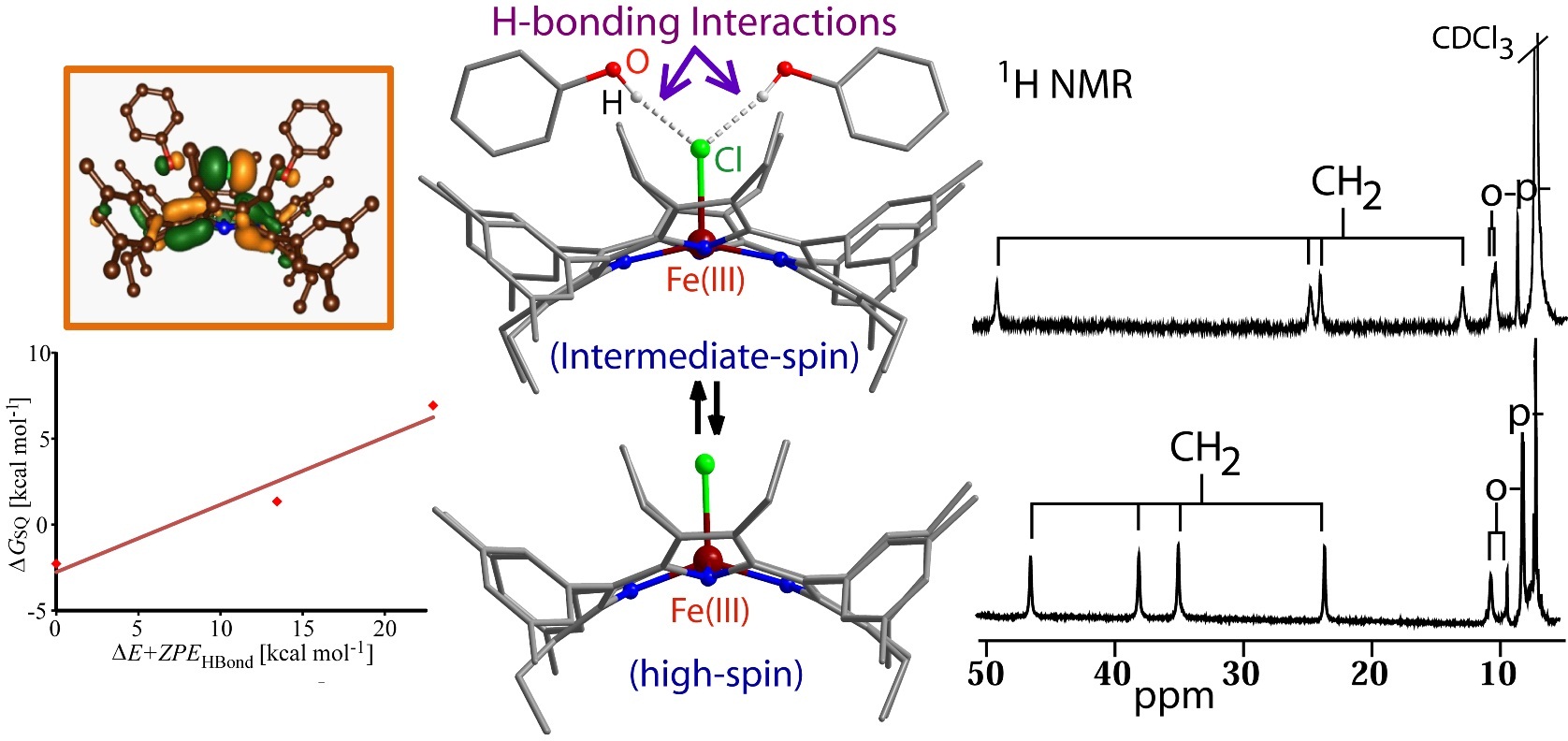
Unlike bis-Fe(IV) state in MauG, the 2e-oxidation has stabilized two ferric hemes, each coupled with a porphyrin cation radical. The extensive π-conjugation enables the generation of a charge resonance band ~1200 nm and allows antiferromagnetic coupling between iron and porphyrin radical spins of both rings. 8. Hydrogen Bonding Interactions Trigger a Spin-Flip in Iron(III)-Porphyrin Complexes D. Sahoo, M. G. Quesne, S. P. de Visser* and S. P. Rath* Angew. Chem. Int. Ed. 2015, 54, 4796-4800. Full Article
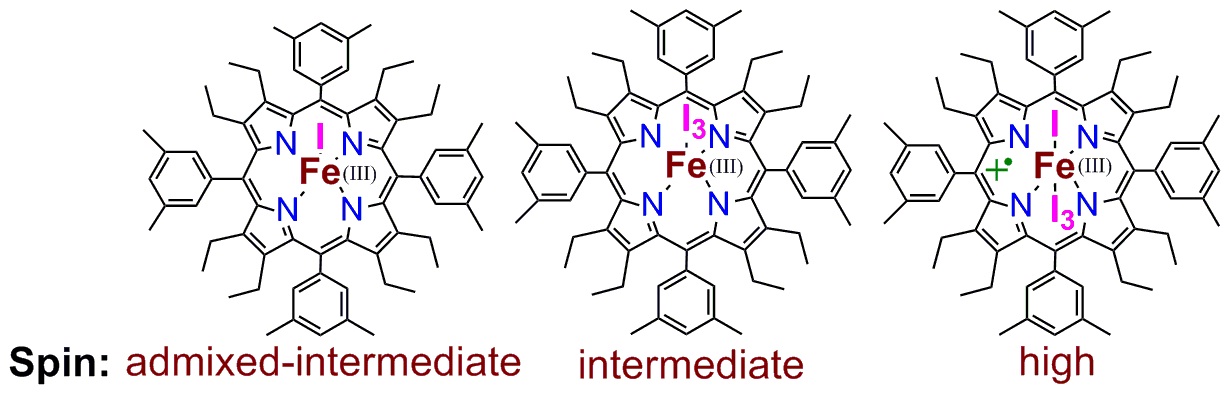
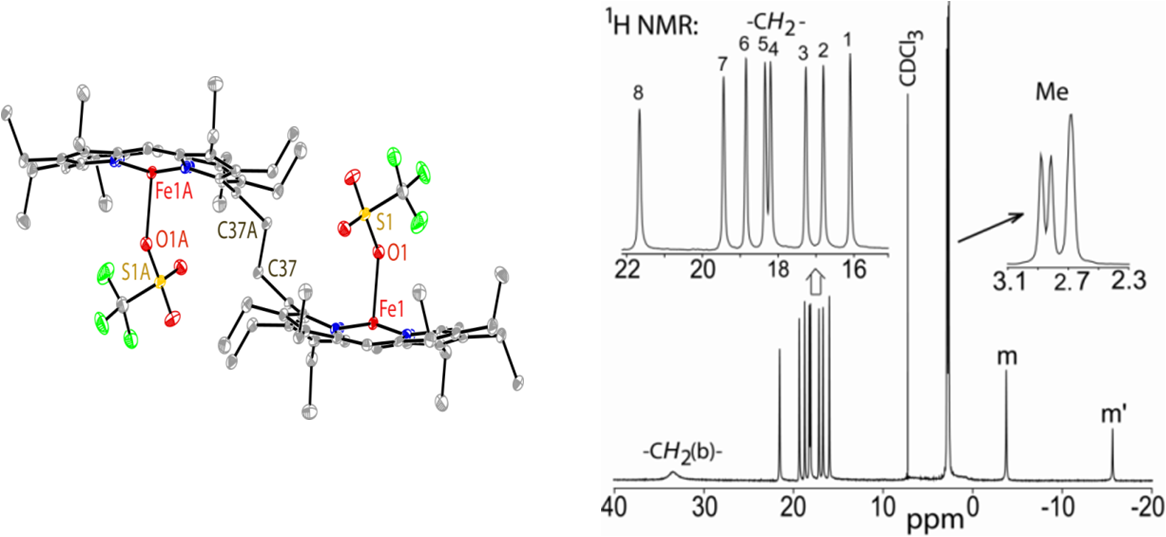
Spin control: The electronic structure of a five-coordinate iron(III) porphinato chloride has been found to depend on the presence of hydrogen-bonding interactions, with the iron center switching reversibly between a high (S=5/2) and intermediate spin (S=3/2) state. Computational calculations clearly support the experimentally assigned spin state. 7. Controlled generation of highly saddled (porphyrinato) iron(III) iodide, tri-iodide and one-electron oxidized complex D. Sahoo and S. P. Rath* Chem. Commun. 2015, 51, 16790-16793. Full Article
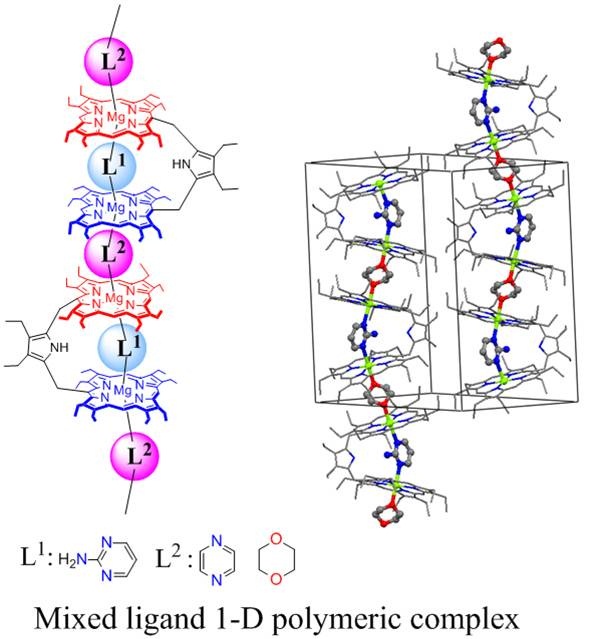
Three iron(III) porphyrinato complexes have been isolated selectively just by varying iodine concentration which eventually form admixed-intermediate (iodo complex), pure intermediate (tri-iodide complex) and high-spin (1e-oxidized complex) states of iron where iodide and/or tri-iodide used as axial ligands. Computational calculations clearly support the experimentally assigned spin state. 6. Metal-coordination-driven Mixed Ligand Binding in Supramolecular Bisporphyrin Tweezers Sk. A. Ikbal, A. Dhamija and S. P. Rath* Chem. Commun. 2015, 51, 14107-14110. Full Article
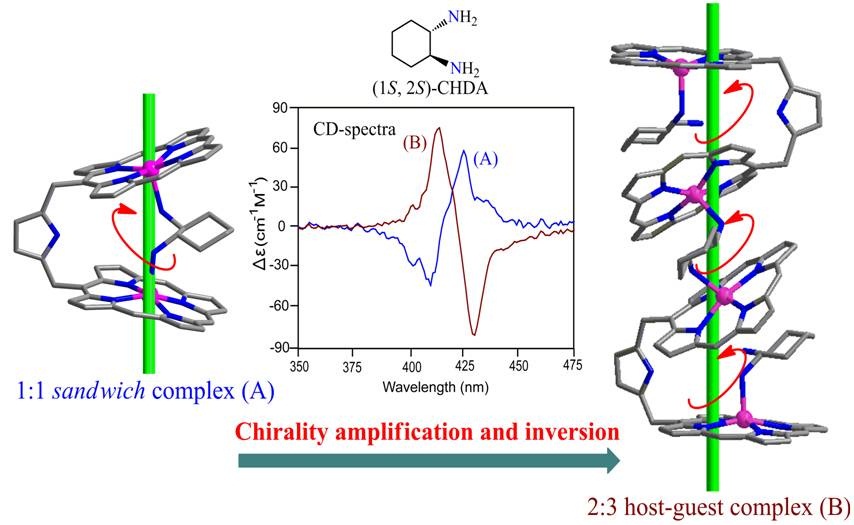
The mixed ligand polymers of Mg(II)bisporphyrin with heterogeneous guest pairs have been synthesized just by mixing host and guests in one pot and structurally characterized. The guest ligands are bound selectively between inside and outside the bisporphyrin cavity. 5. Step-wise induction, amplification and inversion of molecular chirality through the coordination of chiral diamines with Zn(II)bisporphyrin Sk. A. Ikbal, S. Brahma and S. P. Rath* Chem. Commun. 2015, 51, 895-898. Full Article
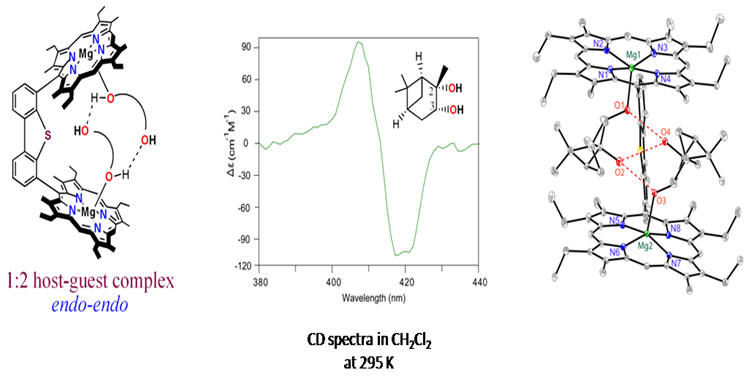
A clear structural rationalization of the origin of chirality transfer from an optically active diamine guest to an achiral Zn(II)bisporphyrin host in a 1:1 and 2:3 host-guest supramolecular complex has been demonstrated for the first time. During the process, chirality inversion along with amplification was observed. 4. Transfer and Control of Molecular Chirality in the 1:2 Host-Guest Supramolecular Complex Consisting of Mg(II) bisporphyrin and Chiral Diols: Effect of H-bonding on Rationalization of Chirality Sk. A. Ikbal, S. Brahma and S. P. Rath* Chem. Commun. 2014, 50, 14037-14040. Full Article
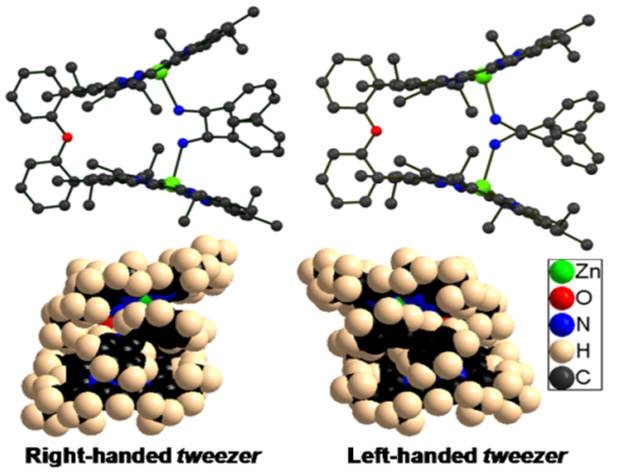
Chiral diol substrate binds to Mg(II)bisporphyrin in an unusual endo-endo fashion due to inter- ligand H-bonding which eventually results in remarkably high amplitude bisignate CD signal. 3. Induction of Supramolecular Chirality in Di-Zinc(II) Bisporphyrin via Tweezer Formation: Synthesis, Structure and Rationalization of Chirality S. Brahma, Sk. A. Ikbal, S. Dey and S. P. Rath* Chem. Commun. 2012, 48, 4070-4072. Full Article (Invited article in a special thematic issue 'Porphyrins and Phthalocyanines')
Two new supramolecular complexes consisting of an achiral bisporphyrin host and a chiral diamine guest are reported. One shows remarkably high amplitude of bisignate CD signal while other one shows very low value. 2. Control of Spins by Ring Deformation in a Diiron(III)bisporphyrin: Reversal of ClO4- and CF3SO3- Ligand Field Strength on the Magnetochemical Series. S. Bhowmik, S. K. Ghosh and S. P. Rath* Chem. Commun. 2011, 47, 4790-4792. Full Article
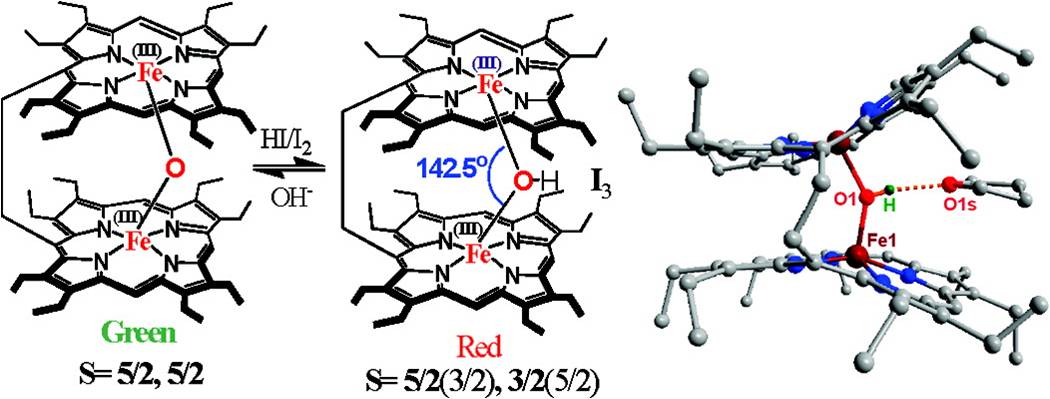
The complete reversal of the ligand field strength between ClO4- and CF3SO3- on the ‘magnetochemical’ series is observed in a diiron(III)bisporphyrin. While ClO4- gives typical high-spin complex, just by twisting the conformation of the macrocycle shows properties with pure intermediate-spin state with CF3SO3- although the axial ligand strengths suggest the reverse order of spin stabilization. 1. A Remarkably Bent Diiron(III)-μ-Hydroxo Bisporphyrin: Unusual Stabilization of Two Spin States of Iron in a Single Molecular Framework S. K. Ghosh and S. P. Rath* J. Am. Chem. Soc. 2010, 132, 17983-17985. Full Article
A novel diiron(III) bisporphyrin bridged by a single hydroxo group between two Fe-centers is reported in which the Fe-O(H)-Fe angle becomes the smallest ever reported in a Fe-porphyrin and two different spin states of Fe are stabilized in a single molecular framework, for the first time, although both cores have exactly the same chemical entity. |
|||||
| ||||||